Results of Comparative Study
Lamberton et al., manuscript in preparation
This study, conducted in Uganda in 2023, aimed to evaluate the performance of POC-CCA3 in the laboratory and field, comparing it to the current POC-CCA and Kato-Katz.
Study setup
- Objective: To evaluate the performance of the POC-CCA3 in comparison to the current POC-CCA and Kato-Katz
- Study setting: School-aged children from a high (Mayuge) and moderate (Tororo) endemic district in Uganda
- Diagnostics: Kato-Katz stool microscopy, POC-CCA and POC-CCA3 with G-score aid to read intensity of infection
Key results
Batch-to-batch variability between the three POC-CCA batches in the study much higher than that between the three POC-CCA3 batches, with the latter showing negligible differences.
Prevalence estimates for intestinal schistosomiasis based on single urine POC-CCA or POC-CCA3 tests was moderately higher than that based on duplicate Kato-Katz (KK) analysis of a single stool sample in a moderate-endemic region in Uganda, while prevalence estimates in a high-endemic region were considerably higher with the POC-CCA and POC-CCA3 compared to KK.
Testing on 3 subsequent days also resulted in higher prevalence estimates with POC-CCA and POC-CCA3 in the moderate-endemicity region, while 3 day duplicate KK based prevalence in the high endemicity region was closer to the estimates based on the two rapid test versions.
Conclusion
The POC-CCA3 is a more consistent and reliable alternative to the POC-CCA, with similar performance in the field, but with improved manufacturing and quality control resulting in a minimal variation between batches.
Study conducted in collaboration with

Other Supporting Data
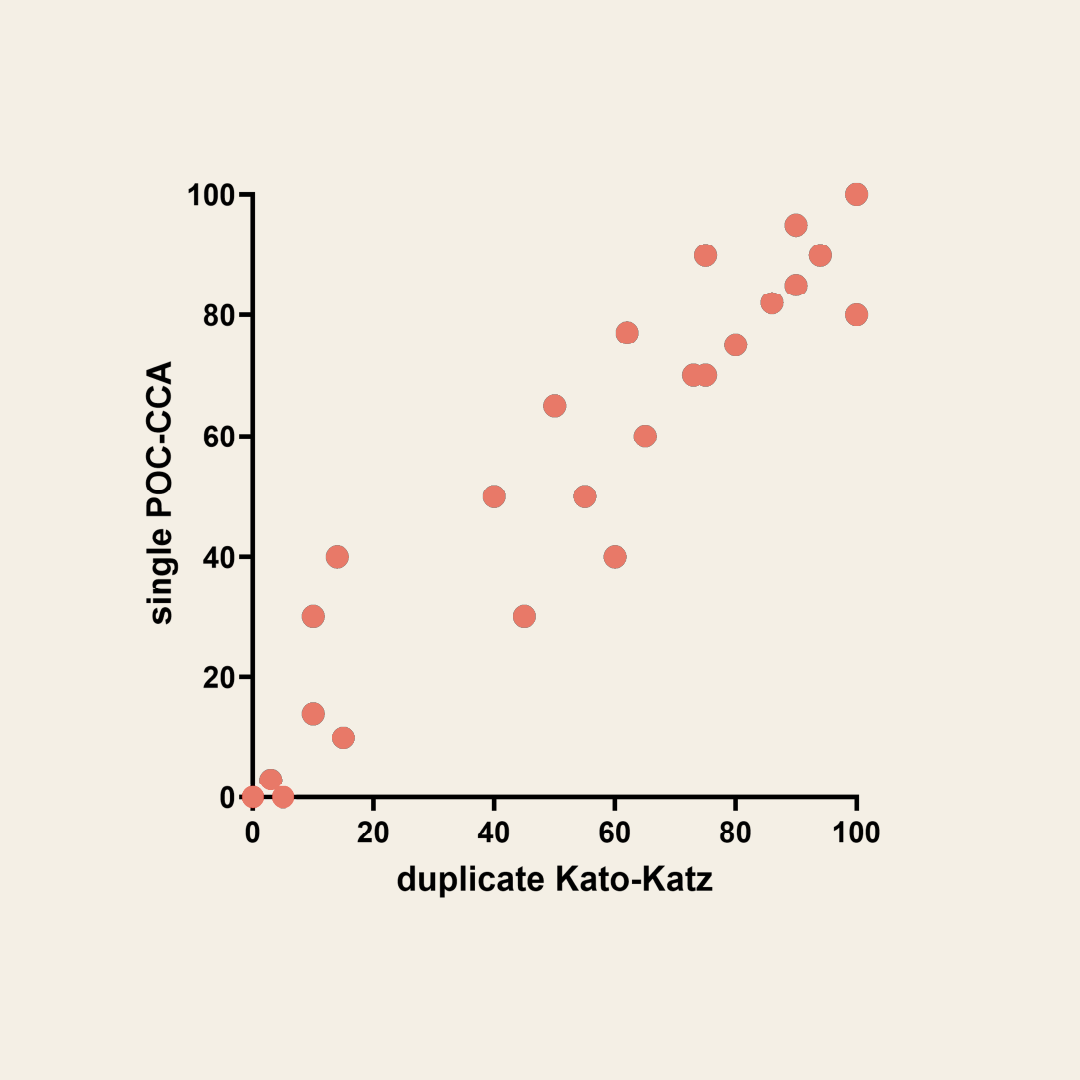
Correlation of POC-CCA and Kato-Katz based prevalence in 25 schools in Uganda. (Based on Stothard et al., 2006 Acta Tropica)
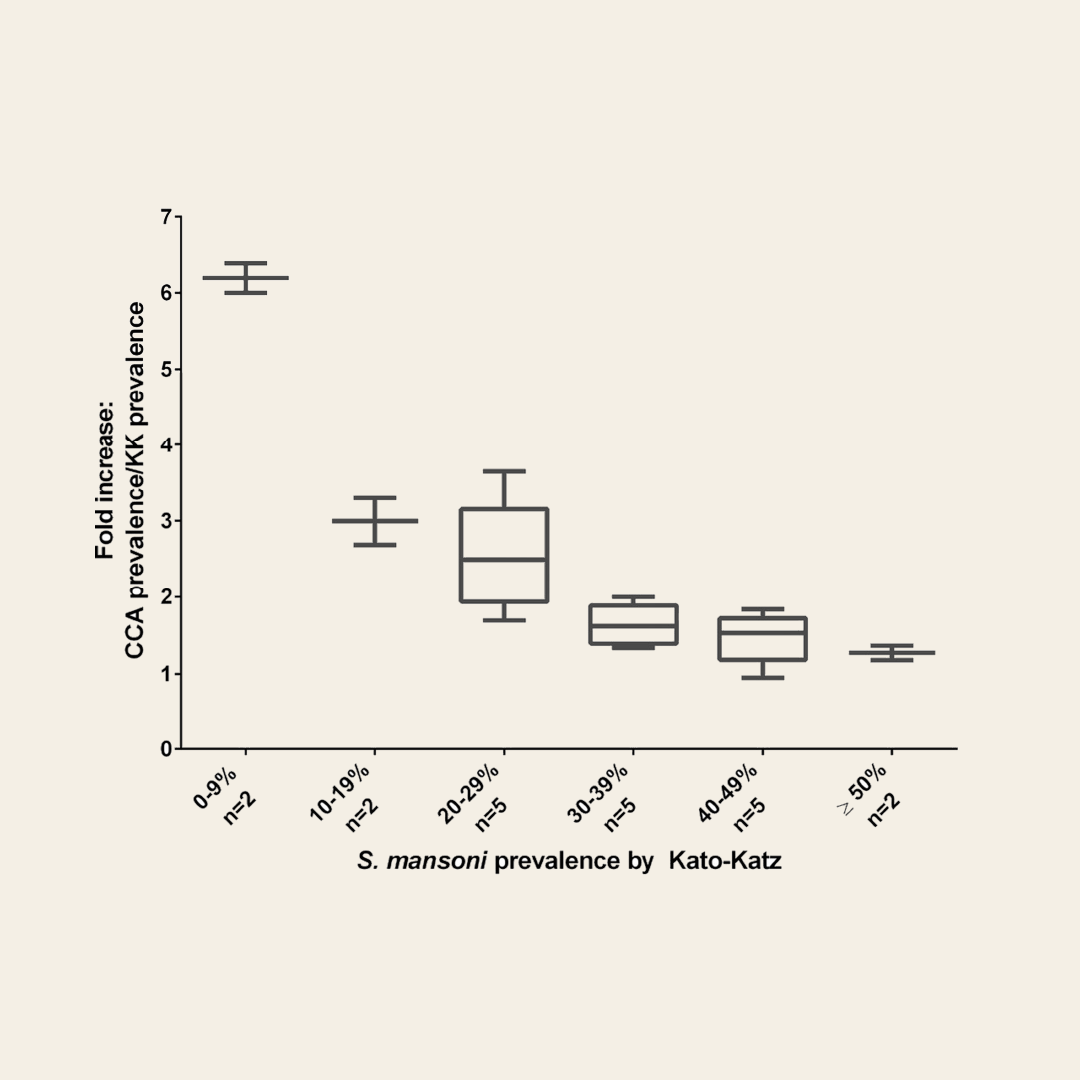
Fold-increase prevalence based on POC-CCA compared to Kato-Katz. (Based on Kittur et al., 2016 AJTMH)
More Updates
Subscribe to The Newsletter
Be the first to know—Get the latest news, discoveries, and impact stories that matter








%204.png)